the 12 principles of green chemistry
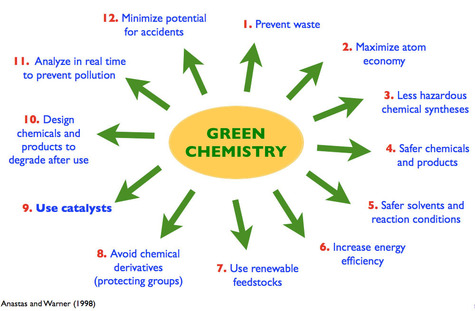
There are 12 main principles of chemistry that Paul
Anastas and John C. Warner developed which
help to explain what the definition means. The principles are as followed, in Anastas' and Warner's own words:
1. It is better to prevent waste than to treat or clean up waste after it has been created.
2. Synthetic methods should be designed to
maximize the incorporation of all materials used in the process into the final product.
3. Wherever practicable, synthetic methods should be designed to use and generate substances
that possess little or no toxicity to human health and the environment.
4. Chemical products should be
designed to effect their desired function while minimizing their toxicity.
5. The use of auxiliary substances (eg. solvents, separation agents, etc.) should be made unnecessary whenever possible and innocuous when used.
6.Energy requirements of chemical processes should be recognized for their environmental and economical impacts and should be minimized. If possible, synthetic methods should be conducted at ambient temperature and pressure.
7. A raw material or feedstock should be renewable rather than depleting whenever technically and economically practicable.
8.Unnecessary derivatization (use of blocking groups, protection/deprotection, temporary modification of physical/chemical processes) should be minimized or avoided if possible, because such steps require additional reagents and can generate waste.
9. Catalytic reagents (as selective as
possible) are superior to stoichiometric reagents.
10.Chemical products should be designed so that at the end of their function they break down into innocuous degradation products and do not persist in the environment.
11. Analytical methodologies need to be further developed to allow for the real-time, in-process monitoring and control prior to the
formation of hazardous substances.
12. Substances and the form of a substance used in a chemical process should be chosen to minimize the potential for chemical
accidents, including releases, explosions, and fires.
(Source: Anastas, P. T. and Warner, J. C. Green Chemistry: Theory and Practice. Oxford
University Press: New York, 1998, p. 30.)
Anastas and John C. Warner developed which
help to explain what the definition means. The principles are as followed, in Anastas' and Warner's own words:
1. It is better to prevent waste than to treat or clean up waste after it has been created.
2. Synthetic methods should be designed to
maximize the incorporation of all materials used in the process into the final product.
3. Wherever practicable, synthetic methods should be designed to use and generate substances
that possess little or no toxicity to human health and the environment.
4. Chemical products should be
designed to effect their desired function while minimizing their toxicity.
5. The use of auxiliary substances (eg. solvents, separation agents, etc.) should be made unnecessary whenever possible and innocuous when used.
6.Energy requirements of chemical processes should be recognized for their environmental and economical impacts and should be minimized. If possible, synthetic methods should be conducted at ambient temperature and pressure.
7. A raw material or feedstock should be renewable rather than depleting whenever technically and economically practicable.
8.Unnecessary derivatization (use of blocking groups, protection/deprotection, temporary modification of physical/chemical processes) should be minimized or avoided if possible, because such steps require additional reagents and can generate waste.
9. Catalytic reagents (as selective as
possible) are superior to stoichiometric reagents.
10.Chemical products should be designed so that at the end of their function they break down into innocuous degradation products and do not persist in the environment.
11. Analytical methodologies need to be further developed to allow for the real-time, in-process monitoring and control prior to the
formation of hazardous substances.
12. Substances and the form of a substance used in a chemical process should be chosen to minimize the potential for chemical
accidents, including releases, explosions, and fires.
(Source: Anastas, P. T. and Warner, J. C. Green Chemistry: Theory and Practice. Oxford
University Press: New York, 1998, p. 30.)

